University of Hawai‘i at Mānoa Food Science and Human Nutrition Program and Human Nutrition Program
Lipids are unique organic compounds, each serving key roles and performing specific functions within the body. As we discuss the various types of lipids (triglycerides, phospholipids, and sterols) in further detail, we will compare their structures and functions and examine their impact on human health.
Triglycerides Structure and Functions
Triglycerides are the main form of lipid found in the body and in the diet. Fatty acids and glycerol are the building blocks of triglycerides. Glycerol is a thick, smooth, syrupy compound that is often used in the food industry. To form a triglyceride, a glycerol molecule is joined by three fatty acid chains. triglycerides contain varying mixtures of fatty acids.
Figure 5.3 The Structure of a Triglycerides
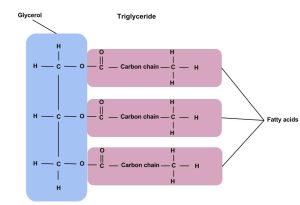
Fatty Acids
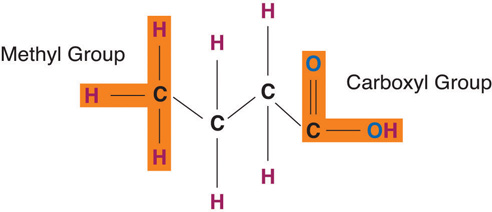
Fatty acids determine if the compound is solid or liquid at room temperature. Fatty acids consist of a carboxylic acid (−COOH) group on one end of a carbon chain and a methyl group (−CH3) on the other end. Fatty acids can differ from one another in two important ways—carbon chain length and degree of saturation.
It’s All in the Chain
Fatty acids have different chain lengths and different compositions. Foods have fatty acids with chain lengths between four and twenty-four carbons and most of them contain an even number of carbon atoms. When the carbon chain length is shorter, the melting point of the fatty acid becomes lower—and the fatty acid becomes more liquid.
Figure 5.4 Structures of a Saturated, Monounsaturated, and Polyunsaturated Fat

Fatty Acid Types in the Body
The fatty-acid profile of the diet directly correlates to the tissue lipid profile of the body. It may not solely be the quantity of dietary fat that matters. More directly, the type of dietary fat ingested has been shown to affect body weight, composition, and metabolism. The fatty acids consumed are often incorporated into the triglycerides within the body. Evidence confirms that saturated fatty acids are linked to higher rates of weight retention when compared to other types of fatty acids. Alternatively, the fatty acids found in fish oil are proven to reduce the rate of weight gain as compared to other fatty acids.[1]
Degrees of Saturation
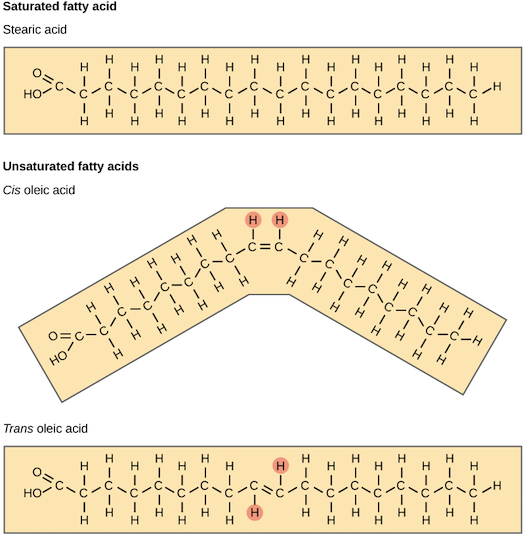
Fatty acid chains are held together by carbon atoms that attach to each other and to hydrogen atoms. The term saturation refers to whether or not a fatty acid chain is filled (or “saturated”) to capacity with hydrogen atoms. If each available carbon bond holds a hydrogen atom we call this a saturated fatty acid chain. All carbon atoms in such a fatty acid chain are bonded with single bonds. Sometimes the chain has a place where hydrogen atoms are missing. This is referred to as the point of unsaturation.
When one or more bonds between carbon atoms are a double bond (C=C), that fatty acid is called an unsaturated fatty acid, as it has one or more points of unsaturation. Any fatty acid that has only one double bond is a monounsaturated fatty acid, an example of which is olive oil (75 percent of its fat is monounsaturated). Monounsaturated fats help regulate blood cholesterol levels, thereby reducing the risk for heart disease and stroke. A polyunsaturated fatty acid is a fatty acid with two or more double bonds or two or more points of unsaturation. Soybean oil contains high amounts of polyunsaturated fatty acids. Both monounsaturated fats and polyunsaturated fats provide nutrition that is essential for normal cell development and healthy skin.
Foods that have a high percentage of saturated fatty acids tend to be solid at room temperature. Examples of these are fats found in chocolate (stearic acid, an eighteen-carbon saturated fatty acid is a primary component) and meat. Foods rich in unsaturated fatty acids, such as olive oil (oleic acid, an eighteen-carbon unsaturated fatty acid, is a major component) tend to be liquid at room temperature. Flaxseed oil is rich in alpha-linolenic acid, which is an unsaturated fatty acid and becomes a thin liquid at room temperature.
Knowing the connection between chain length, degree of saturation, and the state of the fatty acid (solid or liquid) is important for making food choices. If you decide to limit or redirect your intake of fat products, then choosing unsaturated fat is more beneficial than choosing a saturated fat. This choice is easy enough to make because unsaturated fats tend to be liquid at room temperature (for example, olive oil) whereas saturated fats tend to be solid at room temperature (for example, butter). Avocados are rich in unsaturated fats. Most vegetable and fish oils contain high quantities of polyunsaturated fats. Olive oil and canola oil are also rich in monounsaturated fats. Conversely, tropical oils are an exception to this rule in that they are liquid at room temperature yet high in saturated fat. Palm oil (often used in food processing) is highly saturated and has been proven to raise blood cholesterol. Shortening, margarine, and commercially prepared products (in general) report to use only vegetable-derived fats in their processing. But even so, much of the fat they use may be in the saturated and trans fat categories.
Cis or Trans Fatty Acids?
The introduction of a carbon double bond in a carbon chain, as in an unsaturated fatty acid, can result in different structures for the same fatty acid composition. When the hydrogen atoms are bonded to the same side of the carbon chain, it is called a cis fatty acid. Because the hydrogen atoms are on the same side, the carbon chain has a bent structure. Naturally occurring fatty acids usually have a cis configuration.
In a trans fatty acid, the hydrogen atoms are attached on opposite sides of the carbon chain. Unlike cis fatty acids, most trans fatty acids are not found naturally in foods, but are a result of a process called hydrogenation. Hydrogenation is the process of adding hydrogen to the carbon double bonds, thus making the fatty acid saturated (or less unsaturated, in the case of partial hydrogenation). This is how vegetable oils are converted into semisolid fats for use in the manufacturing process.
According to the ongoing Harvard Nurses’ Health Study, trans fatty acids have been associated with increased risk for coronary heart disease because of the way they negatively impact blood cholesterol levels.[2]
Figure 5.5 Structures of Saturated, Unsaturated, Cis and Trans fatty Acids
Interestingly, some naturally occurring trans fats do not pose the same health risks as their artificially engineered counterparts. These trans fats are found in ruminant animals such as cows, sheep, and goats, resulting in trans fatty acids being present in our meat, milk, and other dairy product supply. Reports from the US Department of Agriculture (USDA) indicate that these trans fats comprise 15 to 20 percent of the total trans-fat intake in our diet. While we know that trans fats are not exactly harmless, it seems that any negative effect naturally occurring trans fats have are counteracted by the presence of other fatty acid molecules in these animal products, which work to promote human health.
Learning Activities
Technology Note: The second edition of the Human Nutrition Open Educational Resource (OER) textbook features interactive learning activities. These activities are available in the web-based textbook and not available in the downloadable versions (EPUB, Digital PDF, Print_PDF, or Open Document).
Learning activities may be used across various mobile devices, however, for the best user experience it is strongly recommended that users complete these activities using a desktop or laptop computer and in Google Chrome.
- Mori T, Kondo H. (2007). Dietary fish oil upregulates intestinal lipid metabolism and reduces body weight gain in C57BL/6J mice. Journal of Nutrition, 137(12):2629-34. http://www.ncbi.nlm.nih.gov/pubmed/18029475. Accessed September 22, 2017. ↵
- Introduction to “Fats and Cholesterol: Out with the Bad, In with the Good” The Nutrition Source. Harvard School of Public Health. http://www.hsph.harvard.edu/nutritionsource/what-should-you-eat/fats-full-story/#references. Updated 2017. Accessed September 28, 2017. ↵
A class of nutrients containing carbon, hydrogen, a little oxygen, and some other atoms. Commonly known as fats that include fatty acids, triglycerides, phospholipids, and sterols.
The smallest unit of an element.

