7.2 Enthalpy and Chemical Reactions
Learning Objectives
- Define enthalpy.
- Properly express the enthalpy change of chemical reactions.
- Explain how enthalpy changes are measured experimentally.
Now that we have shown how energy, work, and heat are related, we are ready to consider energy changes in chemical reactions. A fundamental concept is that every chemical reaction occurs with a concurrent change in energy. Now we need to learn how to properly express these energy changes.
Conditions like pressure, volume, and temperature affect the energy content of a system. What we need is a definition of energy that holds when some of these conditions are specified (somewhat similar to our definition of standard temperature and pressure in our study of gases). We define the enthalpy change (ΔH) as the heat of a process when pressure is held constant:
[latex]\Delta H \equiv q\text{ at constant pressure}[/latex]
The letter H stands for “enthalpy,” a kind of energy, while the Δ implies a change in the quantity. We will always be interested in the change in H, rather than the absolute value of H itself.
When a chemical reaction occurs, there is a characteristic change in enthalpy. The enthalpy change for a reaction is typically written after a balanced chemical equation and on the same line. For example, when two moles of hydrogen react with one mole of oxygen to make two moles of water, the characteristic enthalpy change is 570 kJ. We write the equation as:
[latex]\ce{2H2(g)}+\ce{O2(g)}\rightarrow\ce{2H2O(\ell)}\quad \Delta H=-570\text{ kJ}[/latex]
A chemical equation that includes an enthalpy change is called a thermochemical equation. A thermochemical equation is assumed to refer to the equation in molar quantities, which means it must be interpreted in terms of moles, not individual molecules.
Example 7.2
Problem
Write the thermochemical equation for the reaction of PCl3(g) with Cl2(g) to make PCl5(g), which has an enthalpy change of −88 kJ.
Solution
[latex]\ce{PCl3(g)}+\ce{Cl2(g)}\rightarrow \ce{PCl5(g)}\quad \Delta H=-88\text{ kJ}[/latex]
Test Yourself
Write the thermochemical equation for the reaction of N2(g) with O2(g) to make 2NO(g), which has an enthalpy change of 181 kJ.
Answer
[latex]\ce{N2(g)}+\ce{O2(g)}\rightarrow \ce{2NO(g)}\quad \Delta H = 181\text{ kJ}[/latex]
You may have noticed that the ΔH for a chemical reaction may be positive or negative. The number is assumed to be positive if it has no sign; a + sign can be added explicitly to avoid confusion. A chemical reaction that has a positive ΔH is said to be endothermic, while a chemical reaction that has a negative ΔH is said to be exothermic.
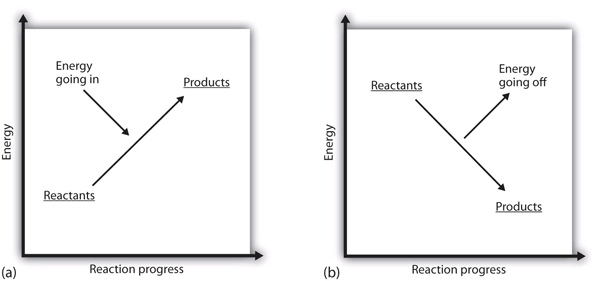
What does it mean if the ΔH of a process is positive? It means that the system in which the chemical reaction is occurring is gaining energy. If one considers the energy of a system as being represented as a height on a vertical energy plot, the enthalpy change that accompanies the reaction can be diagrammed as in part (a) in Figure 7.1 “Reaction Energy”: the energy of the reactants has some energy, and the system increases its energy as it goes to products. The products are higher on the vertical scale than the reactants. Endothermic, then, implies that the system gains, or absorbs, energy.
An opposite situation exists for an exothermic process, as shown in part (b) in Figure 7.3 “Reaction Energy.” If the enthalpy change of a reaction is negative, the system is losing energy, so the products have less energy than the reactants, and the products are lower on the vertical energy scale than the reactants are. Exothermic, then, implies that the system loses, or gives off, energy.
Example 7.3
Problem
Consider this thermochemical equation:
[latex]\ce{2CO(g)}+\ce{O2(g)}\rightarrow \ce{2CO2(g)}\quad \Delta H = -565\text{ kJ}[/latex]
Is it exothermic or endothermic? How much energy is given off or absorbed?
Solution
By definition, a chemical reaction that has a negative ΔH is exothermic, meaning that this much energy — in this case, 565 kJ — is given off by the reaction.
Test Yourself
Consider this thermochemical equation:
[latex]\ce{CO2(g)}+\ce{H2(g)}\rightarrow \ce{CO(g)}+\ce{H2O(g)}\quad \Delta H=42\text{ kJ}[/latex]
Is it exothermic or endothermic? How much energy is given off or absorbed?
Answer
Endothermic; 42 kJ are absorbed.
Key Takeaways
- Every chemical reaction occurs with a concurrent change in energy.
- The change in enthalpy equals heat at constant pressure.
- Enthalpy changes can be expressed by using thermochemical equations.
Exercises
Questions
- Under what circumstances are q and ΔH the same?
- Under what circumstances are q and ΔH different?
- Hydrogen gas and chlorine gas react to make hydrogen chloride gas with an accompanying enthalpy change of −184 kJ. Write a properly balanced thermochemical equation for this process.
- Propane (C3H8) reacts with elemental oxygen gas to produce carbon dioxide and liquid water with an accompanying enthalpy change of −2,220 kJ. Write a properly balanced thermochemical equation for this process.
- Nitrogen gas reacts with oxygen gas to make NO(g) while absorbing 180 kJ. Write a properly balanced thermochemical equation for this process.
- Solid sodium reacts with chlorine gas to make solid sodium chloride while giving off 772 kJ. Write a properly balanced thermochemical equation for this process.
- Hydrogen gas and chlorine gas react to make hydrogen chloride gas with an accompanying enthalpy change of −184 kJ. Is this process endothermic or exothermic?
- Propane (C3H8) reacts with elemental oxygen gas to produce carbon dioxide while giving off 2,220 kJ of energy. Is this process endothermic or exothermic?
- Nitrogen gas reacts with oxygen gas to make NO(g) while absorbing 180 kJ. Is this process exothermic or endothermic?
- Sodium metal can react with nitrogen to make sodium azide (NaN3) with a ΔH of 21.72 kJ. Is this process exothermic or endothermic?
- Draw an energy level diagram for the chemical reaction in Exercise 8. (See Figure 7.3 “Reaction Energy” for an example.)
- Draw an energy level diagram for the chemical reaction in Exercise 9. (See Figure 7.3 “Reaction Energy” for an example.)
- In a 250 mL solution, 0.25 mol of KOH(aq) and 0.25 mol of HNO3(aq) are combined. The temperature of the solution increases from 22.5°C to 35.9°C. Assume the solution has the same density and heat capacity of water. What is the heat of the reaction, and what is the ΔH of the reaction on a molar basis?
- In a 600 mL solution, 0.50 mol of Ca(OH)2(aq) and 0.50 mol of H2SO4(aq) are combined. The temperature of the solution increases by 22.3°C. What is the heat of the reaction, and what is the ΔH of the reaction on a molar basis? Assume the solution has the same density and heat capacity of water.
- To warm 400.0 g of H2O, 0.050 mol of ethanol (C2H5OH) is burned. The water warms from 24.6°C to 65.6°C. What is the heat of the reaction, and what is the ΔH of the reaction on a molar basis?
- To warm 100.0 g of H2O, 0.066 mol beeswax is burned. The water warms from 21.4°C to 25.5°C. What is the heat of the reaction, and what is the ΔH of the reaction on a molar basis?
Answers
- under conditions of constant pressure
- [latex]\ce{H2(g)}+\ce{Cl2(g)}\rightarrow\ce{2HCl(g)}\quad \Delta H=-184\text{ kJ}[/latex]
- [latex]\ce{N2(g)}+\ce{O2(g)}\rightarrow \ce{2NO(g)}\quad \Delta H=180\text{ kJ}[/latex]
- exothermic
- endothermic
- [latex][/latex]\setlength{\unitlength}{1cm}
- heat of reaction = −14.0 kJ; ΔH = −56.0 kJ/mol of reactants
- heat of reaction = −68.6 kJ; ΔH = −1,370 kJ/mole of ethanol
Media Attributions
- "Reaction Energy" by David W. Ball © CC BY-NC-SA (Attribution-NonCommercial-ShareAlike)
- "Calorimeter" by David W. Ball © CC BY-NC-SA (Attribution-NonCommercial-ShareAlike)

